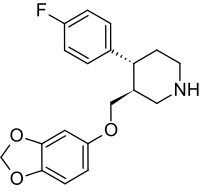
Paroxetine, also known by the trade names Paxil and Seroxat.
– Pivotal Lawsuit Shows Widely Used Antidepressant Increases Suicide Risk Over 800 Percent:
A trial is currently underway in Illinois as a widow seeks to hold pharmaceutical manufacturer GlaxoSmithKline accountable for improper labeling and minimizing a potentially serious side effect of a well-known antidepressant.
Paroxetine is a widely prescribed antidepressant and anti-anxiety drug under the class of drugs known as selective serotonin reuptake inhibitors (SSRIs). Paroxetine is most commonly known as the brand name of Paxil, manufactured by GlaxoSmithKline (GSK).
…
H/t reader kevin a.
Related info:
– Antidepressant Paxil (6.1% Fluoride!) Promotes Breast Cancer
Antidepressants, like Paxil, cause all kinds of cancer …
Paroxetine (Paxil)
… because like with Prozac …
… the active ingredient is fluoride!
And …
“Fluoride causes more human cancer, and causes it faster, than any other chemical.”
– Dean Burk, Chief Chemist Emeritus, US National Cancer Institute“Yes, Prozac is 18.4% fluoride, by molecular weight. Luvox is 17.9% fluoride. Paxil is 6.1% fluoride. All these drugs numb your mind, using fluoride as the key element that reacts with your brain tissue.“
Source: Mike Adams, Natural News
Cancer is just one among many detrimental health effects caused by fluoride.
* * *
PayPal: Donate in USD
PayPal: Donate in EUR
PayPal: Donate in GBP